How Salt, Microbes, and Time Preserved Food
Before refrigeration, preserving food was a survival necessity. Salt, microbes, and time worked together to prevent spoilage and extend the shelf life of perishable items. Here's how it worked:
- Salt: Draws out moisture through osmosis, inhibiting bacteria growth.
- Microbes: Beneficial bacteria, like lactic acid bacteria, thrive in salty environments, producing acids that lower pH and prevent harmful pathogens.
- Time: Allows preservation processes to fully develop, enhancing safety and flavor.
This combination enabled ancient civilizations to store food for months, settle in one place, and even undertake long journeys. Methods like salting, brining, and fermentation were widely used, shaping culinary practices still relevant today. Modern research confirms the science behind these methods, offering safe ways to recreate them at home.
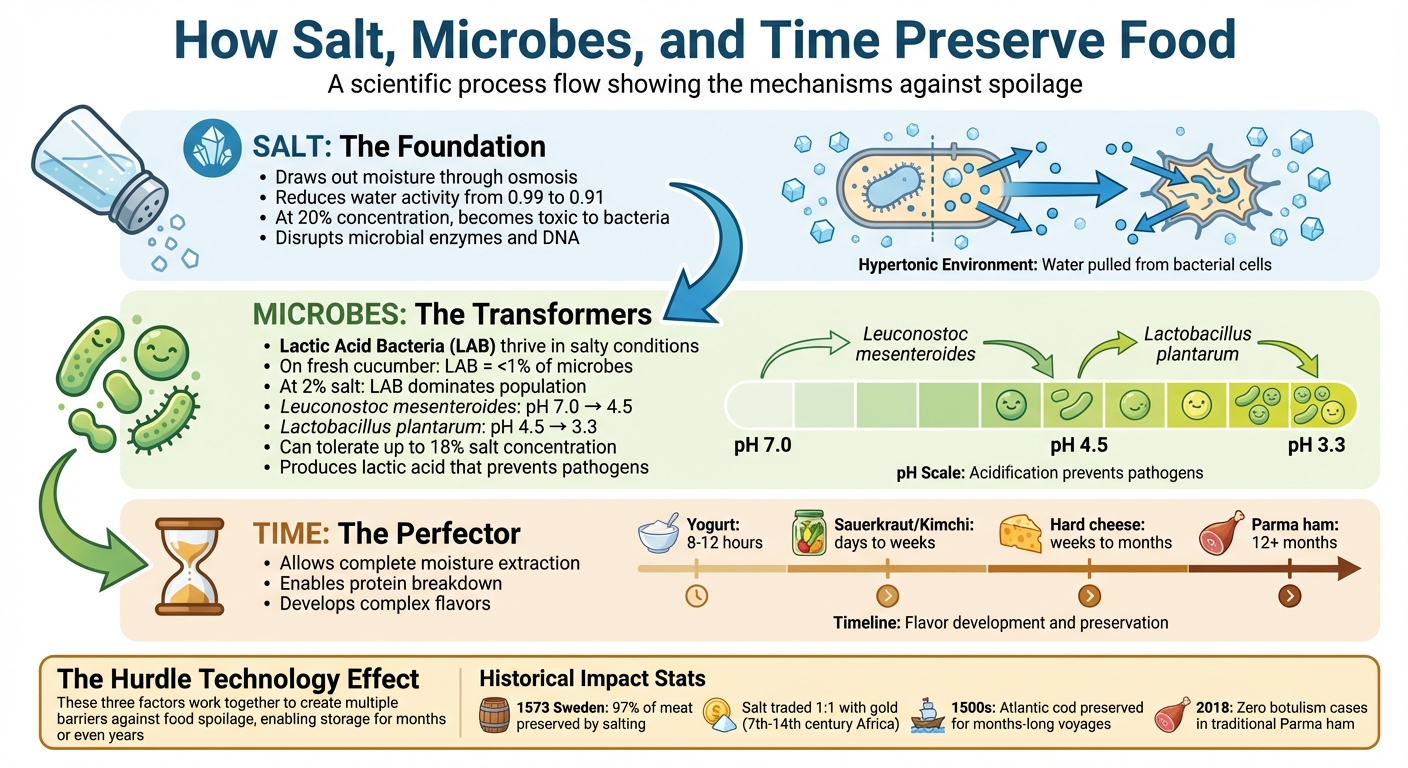
How Salt, Microbes, and Time Preserve Food: A Scientific Process
How Does Salt Preserve Food? [ID0913]
sbb-itb-4066b8e
How Salt Preserves Food
Salt works as a powerful preservative by halting the growth and reproduction of harmful bacteria. When applied to foods like meat, fish, or vegetables, salt draws water out of both the food and any microorganisms present through osmosis. Laurie L. Dove puts it this way:
"Salt inhibits bacteria in a variety of ways. It's a disrupter that disrupts microbial enzymes and damages their DNA."
The science behind this process is straightforward. Salt binds to water molecules, reducing the amount of free water available for bacteria to thrive. Fresh foods typically have a water activity level of around 0.99, but lowering it to about 0.91 is usually enough to stop most bacterial growth. Additionally, when salt concentrations reach nearly 20%, the salt itself becomes toxic to many bacteria, disrupting their internal functions .
Next, let’s look at how ancient societies used these properties to preserve food.
Ancient Salt Preservation Practices
Long before refrigeration, ancient civilizations relied on salt's ability to preserve food. The Romans, for example, stored fish in terracotta urns packed with salt. The Greeks created tarichos - salt-cured meat and fish - while the Gauls became known for exporting salted pork and sausages to Rome. In China, the Compendium of Materia Medica documented over 40 types of salt and their antiseptic uses.
Different cultures developed unique methods of salting based on their needs and resources. Dry salting, where salt crystals were rubbed directly onto foods like bacon, venison, cod, and herring, was widely practiced. Brining, which involved soaking food in a saltwater solution, was commonly used for preserving vegetables like cabbage and beans, or creating a traditional aged balsamic. Another method, corning, used coarse salt pellets (referred to as "corns") to rub into meat and prevent spoilage . In 14th-century Europe, venison was first boiled in brine and then dry-salted in barrels to ensure long-term storage.
Salt was so valuable that it became a form of currency in some societies. Roman soldiers were paid a salarium, a wage specifically for purchasing salt. Between the 7th and 14th centuries in western Africa, salt was sometimes traded on a one-to-one weight basis with gold. Historical records from 1573 show that servants of King Gustavus Vasa of Sweden consumed 102 kg of beef and pork, with 99 kg (about 97%) preserved through salting and drying.
These ancient practices were grounded in the scientific principles of osmosis, which we’ll explore next.
How Osmosis Works in Salt Preservation
Osmosis is the driving force behind salt's effectiveness as a preservative. When salt is applied to food, it creates a hypertonic environment that pulls water from microbial cells, dehydrating and disabling them .
High salt concentrations also disrupt enzymes in both microbes and food, preventing spoilage and maintaining the texture and color of the preserved items. Christy Clutter, Ph.D., from the American Society for Microbiology, explains it well:
"While not all microbes are killed by high salinity, many potential pathogens die by osmosis as the salt draws all the water out of them. Others simply find it too energetically costly to survive."
This process not only slowed spoilage but also enabled food to be stored for extended periods. For example, European traders in the 1500s used this technique to preserve large quantities of Atlantic cod from the Grand Banks off Newfoundland. The salted cod could then be transported back to Europe without spoiling. Similarly, North Africans and Indians used salt to preserve lemons for up to a year. Even today, traditional Parma ham relies solely on salting techniques. Since 1993, authentic Parma ham has been made without nitrites, and a 2018 report confirmed zero cases of botulism, despite the absence of chemical preservatives.
Microbes in Food Preservation
Salt does more than just ward off harmful bacteria - it sets the stage for beneficial microbes to flourish. These microorganisms thrive in salty conditions and play a key role in fermentation. Salt essentially acts as a gatekeeper, holding back spoilage organisms while encouraging salt-tolerant lactic acid bacteria (LAB) to grow. This process is a prime example of "hurdle technology", where salt, microbes, and time combine to create multiple layers of protection against food spoilage.
On a fresh cucumber, LAB make up less than 1% of the total microbial population, but salt dramatically shifts this balance. At around 2% salt concentration, most spoilage organisms are suppressed, while salt-tolerant LAB multiply. These bacteria feed on sugars and produce lactic acid, which lowers the pH and further discourages harmful pathogens. According to the Institute of Medicine:
"Salt favors the growth of these more salt-tolerant, beneficial organisms while inhibiting the growth of undesirable spoilage bacteria and fungi naturally present in these foods."
Salt and Lactic Acid Fermentation
Lactic acid fermentation unfolds in stages, driven by different bacteria. The process usually begins with Leuconostoc mesenteroides, which reduces the pH from 7.0 to about 4.5. As the environment becomes more acidic, Lactobacillus plantarum takes over, pushing the pH down to as low as 3.3. Impressively, L. plantarum can handle salt concentrations as high as 18% in some instances.
Salt not only selects for these microbes but also creates the perfect conditions for them to thrive. When salt is added to vegetables, it draws out water and sugars through osmosis, creating a brine. This brine fills air pockets, creating an oxygen-free environment where LAB can dominate the fermentation process. The Exploratorium highlights this process:
"Adding salt to your pickling brine is one important way to help lactic acid bacteria win the microbial race. At a certain salt concentration, lactic acid bacteria grow more quickly than other microbes, and have a competitive advantage."
The exact salt concentration plays a crucial role in shaping the microbial environment during fermentation.
How Salt Controls Microbial Growth
Salt concentration is a key factor in managing microbial activity. At levels of 1.5% or higher, salt can prevent Clostridium botulinum from producing toxins for at least 42 days. Higher concentrations, around 10%, slow fermentation but ensure firmer textures and reduce the risk of spoilage.
Salt also disrupts microbial enzymes and forces bacteria to expend energy on expelling sodium ions. As professional cook and writer Tim Chin points out:
"Salt inhibits growth of many microorganisms through osmotic shock, effectively killing those microbes."
This creates a kind of microbial obstacle course, where only specific, desirable organisms can thrive. For instance, pathogens struggle to survive in low-salt environments, but beneficial bacteria like Pediococcus can handle salt levels up to 8%, continuing to preserve food even under challenging conditions.
The Role of Time in Preservation
Time plays a crucial part in perfecting preserved foods, building on the groundwork laid by salt's osmosis and microbes' fermentation. While salt and microbes kickstart the preservation process, time allows the transformation to fully unfold. Without enough time, moisture extraction remains incomplete, proteins aren't thoroughly broken down, and beneficial bacteria can't finish their work. As the National Center for Home Food Preservation puts it:
"cured meat flavor is thought to be a composite result of the flavors of the curing agents and those developed by bacterial and enzymatic action".
How Aging Develops Flavor
Extended aging is what turns salted foods into flavor powerhouses. Take Parma ham, for instance - it needs at least 12 months of aging. During this time, salt interacts with the meat's pigments, and enzymes break proteins into aromatic compounds.
Cheese undergoes a similar transformation. Christy Clutter, Ph.D., from the American Society for Microbiology, explains:
"Over the course of days, weeks and even months, the storage conditions of different cheeses cultivate unique microbial ecosystems that blossom into the distinct and sometimes pungent flavors we recognize".
This shows how aging elevates flavor complexity. For example, yogurt ferments within 8 to 12 hours, but hard cheeses and dry-cured meats take weeks or even months to reach their peak. Over these extended periods, bacteria produce secondary compounds like esters, which add fruity notes, and diacetyl, which brings creamy flavors. Simultaneously, moisture loss intensifies taste and firms up textures.
But flavor development isn't just about waiting - it requires precise environmental conditions.
Environmental Controls for Long-Term Storage
Time alone can’t do the job; it's the combination of time with controlled temperature, humidity, and atmosphere that ensures success. In the 1980s, New Zealand lamb exporters pioneered controlled atmosphere techniques, which extended fresh meat's shelf life to 12–16 weeks. This innovation allowed lamb to be shipped by boat to Europe and sold as "chilled" rather than frozen.
Carefully managed conditions slow microbial growth and enhance enzymatic activity. For instance, canning low-acid foods demands temperatures around 250°F (121°C) under pressure to kill heat-resistant Clostridium botulinum spores. Techniques like smoking or drying create surface dehydration, forming a protective barrier that resists bacterial invasion. In cold smoking, quick initial drying is especially critical to curb bacterial growth until salt and smoke stabilize the meat.
Modern preservation often uses "hurdle technology", combining factors like salt levels, low temperatures, reduced pH, and controlled atmospheres to ensure both safety and optimal flavor.
Historical Methods in Modern Practice
Ancient preservation techniques, rooted in the use of salt, microbes, and time, have evolved through modern scientific advancements. These methods, once essential for survival during long journeys, now play a significant role in both industrial food production and home cooking. Modern research has not only validated these techniques but also refined them for improved safety and efficiency.
Scientific Research on Preservation
Modern food science has shed light on the science behind many traditional preservation methods. Take "bay salt", for example - solar-evaporated sea salt that naturally contains microbes capable of producing nitrates and nitrites. These compounds enhance the flavor and color of cured meats while also improving their preservation. This discovery eventually led to the creation of Prague powder, a curing salt with 6.25% sodium nitrite. It ensures consistent safety and gives cured meats their signature pink color .
Salt's ability to reduce water activity through osmotic shock - a principle confirmed by modern research - forms the foundation of hurdle technology. This approach combines salt, controlled pH, and temperature management to ensure food safety while allowing for reduced sodium levels compared to traditional methods . For instance, in 2004, Kraft Foods used predictive modeling to develop "no-growth" formulations for cured meats. By combining salt with lactate and diacetate, they effectively controlled Listeria monocytogenes while lowering sodium content.
Today’s curing processes also optimize nitrite levels for safety and incorporate antioxidants like Vitamin C to prevent the formation of harmful nitrosamines.
Using Ancient Techniques at Home
Thanks to modern research, home cooks can safely adapt these time-tested methods. For example, yogurt can be fermented in 8–12 hours using Lactobacillus, while sauerkraut and kimchi rely on lactic acid fermentation for both preservation and flavor. When canning, maintaining a pH below 4.6 by adding lemon juice or using a pressure canner at around 250°F is essential to prevent the growth of Clostridium botulinum .
Dry-curing meats at home typically requires salt concentrations of up to 20% to eliminate harmful bacteria . Adding a touch of sugar - whether from honey, maple syrup, or corn syrup - can balance the saltiness without compromising preservation. Modern electric dehydrators replicate ancient sun-drying techniques by controlling temperature and airflow, ensuring consistent drying while reducing spoilage . Additionally, when pickling, using glass or stoneware containers helps avoid unwanted chemical reactions between acids and metal.
A great example of traditional methods thriving in modern times is Parma ham. Since 1993, authentic Parma ham has been made using only salt, without nitrites or nitrates. This demonstrates that with strict environmental controls, traditional techniques can produce safe, high-quality results.
These practices honor the historical interplay of salt, microbes, and time, bridging ancient wisdom with today’s culinary advancements. Big Horn Olive Oil embraces these methods, delivering products that reflect the rich tradition and quality of preserved foods.
Conclusion
Preserving food using salt, microbes, and time stands as one of humanity's lasting accomplishments. Salt plays a key role by lowering water activity and drawing moisture out of harmful bacteria through osmosis. Meanwhile, beneficial microbes - particularly Lactic Acid Bacteria - take center stage by transforming sugars into lactic acid. This process reduces pH levels, creating an environment that halts the growth of pathogens. Over time, these microbial interactions not only ensure safety but also produce the complex flavors found in aged cheeses, cured meats, and fermented vegetables.
These age-old techniques still hold relevance today, enriching modern kitchens while keeping us connected to long-standing traditions. As Brian A. Nummer, Ph.D., from the National Center for Home Food Preservation, explains:
"Interests have shifted from preserve 'because we have to', to 'preserve because we like to'".
What was once a necessity for survival has evolved into a way to enhance culinary experiences.
Modern research continues to affirm the effectiveness of these traditional practices. Whether it’s fermenting yogurt or aging cheese, the same core principles apply. By blending ancient techniques with contemporary knowledge, both home cooks and artisans can create preserved foods that honor the past while meeting today’s expectations for safety and flavor. This harmony of tradition and progress not only celebrates our culinary heritage but also reflects the dedication at Big Horn Olive Oil to produce goods of enduring excellence.
FAQs
What salt level is safest for home fermentation?
For safe home fermentation, it's best to stick with salt levels between 2-10% by weight. For vegetables, a range of 2-3% is most common. This amount helps keep harmful bacteria at bay while supporting the growth of lactic acid bacteria, which are key to the fermentation process.
How do I know a ferment is safe to eat?
A safe fermentation process will display clear signs such as a tangy, pleasant smell, visible bubbling, and a soft texture. However, if you notice any mold, sliminess, foul odors, or unusual discoloration, these are red flags that the ferment has spoiled and should not be consumed. To keep your ferment safe, it's crucial to maintain proper salt levels, ensure adequate acidity, and create a controlled environment.
Why do cured foods need weeks or months to age?
Cured foods need weeks or even months to age because this period lets salt do its job: stopping harmful microbes, pulling out moisture, and encouraging the growth of good bacteria. These bacteria create acids that not only help preserve the food but also deepen its flavor. This slow process is key to ensuring both safety and the creation of rich, layered tastes.

